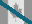Gemcitabine HCl
DNA polymerase inhibitor / Gemcitabine is a clinically useful anticancer agent.1 It exerts its cytotoxic effects via the metabolites gemcitabine diphosphate (dFdCDP) and gemcitabine triphosphate (dFdCTP). dFdCTP is an inhibitor of DNA polymerase and is also incorporated in DNA strands resulting in termination of chain elongation and apoptosis. dFdCDP is an inhibitor of ribonucleotide reductase which results in depletion of deoxyribonucleotides needed for DNA synthesis. Gemcitabine metabolites have also been reported to inhibit cytidine triphosphate synthetase (CTP synthetase)2 and deoxycytidylate deaminase (dCMP deaminase)3. Topoisomerase 1 has also been shown to be a target for gemcitabine.4
Biochemicals & reagents
122111-03-9
dFdC
1) Mini et al. (2006), Cellular Pharmacology of Gemcitabine; Ann.Oncol. 17 v7 2) Heinemann et al. (1995), Gemcitabine: a modulator of intracellular nucleotide and deoxynucleotide metabolism; Semin.Oncol. 22 11 3) Heinemann et al. (1992), Cellular elimination of 2’,2’-difluorodeoxycytidine 5’triphosphate: a mechanism of self-potentiation; Cancer Res. 52 533 4) Pourquier et al. (2002), Gemcitabine (2’,2’-difluoro-2’-deoxycytidine), an antimetabolite that poisons topoisomerase I; Clin.Cancer Res. 8 2499
-20°C
TARGET: DNA -- PATHWAY: DNA synthesis; Apoptosis inducer; Cell cycle; Cytotoxicity; Cytokine -- RESEARCH AREA: Cell death; Immunology -- DISEASE AREA: Inflammation; Cancer